Periodic Table |
 |
 |
 |
 |
 |
 |
 |
| What is the Periodic Table Showing? | Periodicity |
The INTERNET Database of Periodic Tables
There are thousands of periodic tables in web space, but this is the only comprehensive database of periodic tables & periodic system formulations. If you know of an interesting periodic table that is missing, please contact the database curator: Mark R. Leach Ph.D. The database holds information on periodic tables, the discovery of the elements, the elucidation of atomic weights and the discovery of atomic structure (and much, much more).
Periodic Tables from the year 2004:
| Year: 2004 | PT id = 46, Type = misc |
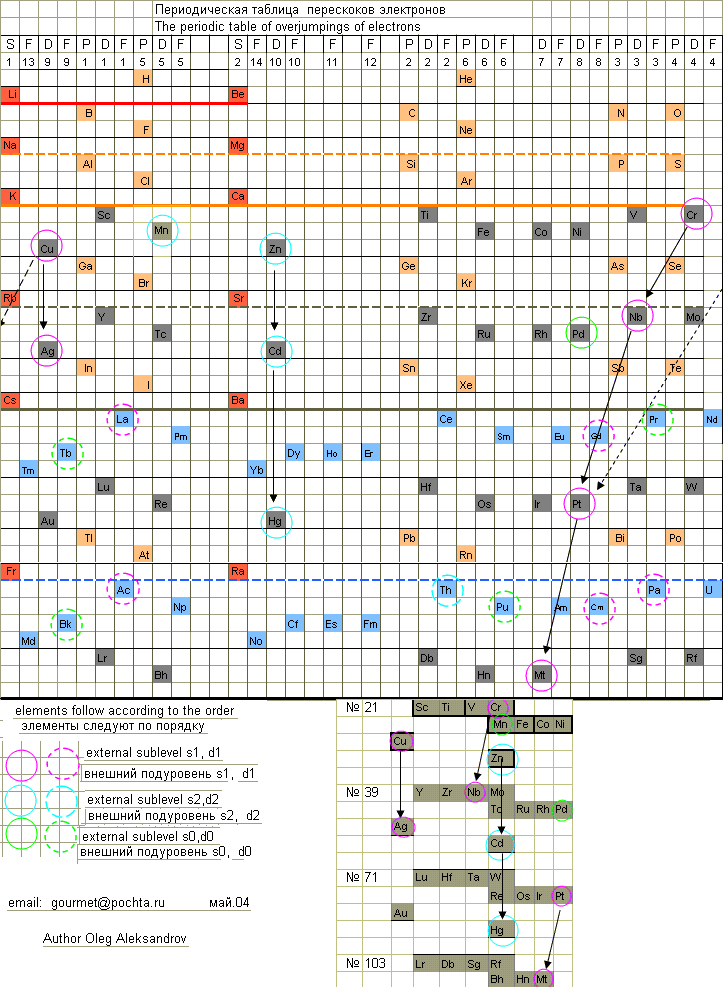
Electron Overjump Periodic Table
Here are some origional periodic table ideas, including history and electron overjumpings by Oleg Aleksandrov, from here.
| Year: 2004 | PT id = 48, Type = formulation 3D spiral |
Rafael Poza Periodic Table (Click to Enlarge)
| Year: 2004 | PT id = 94, Type = non-chem |
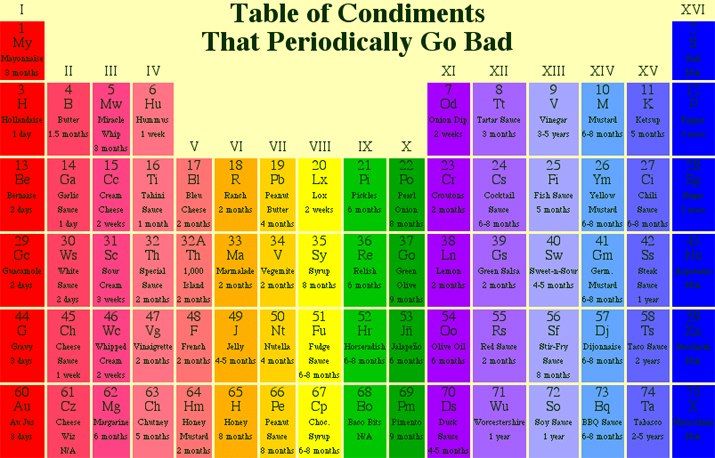
Condiments
A periodic table of condiment lifetimes:
| Year: 2004 | PT id = 111, Type = review |
Rouvray & King's The Periodic Table: Into the 21st Century
D. H Rouvray and R. B. King (ed.), The Periodic Table: Into the 21st Century, Research Studies Press 2004.
| Year: 2004 | PT id = 112, Type = misc |
Periodic Tables in Two Hundred Languages
Periodic Tables of the Elements in Two Hundred Languages:
| Year: 2004 | PT id = 113, Type = review |
Peter van der Krogt's Elementymology & Elements Multidict
Peter van der Krogt's Elementymology & Elements Multidict, the web site for element names, origins (etymology) of element names and translations into other languages.
| Year: 2004 | PT id = 115, Type = data |
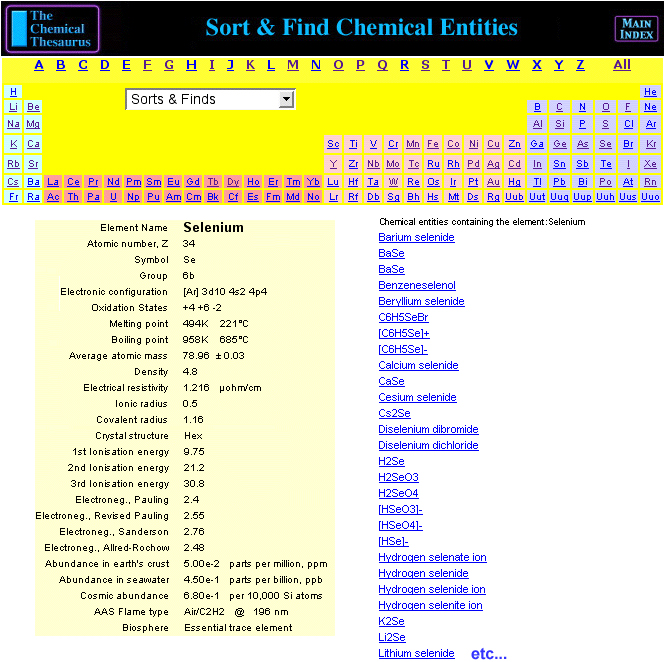
Chemical Thesaurus Periodic Table
Search for chemical reagents, atomic and molecular ions, minerals, isotopes, elemental data, etc., using the periodic table built into The Chemical Thesaurus reaction chemistry database:
By Mark Leach
| Year: 2004 | PT id = 117, Type = data |
Visual Elements Periodic Table
Visual Elements Periodic Table
| Year: 2004 | PT id = 124, Type = data |
Material Type Periodic Table
All of the the main group elements are common laboratory reagents or chemical in bottles. They appear as metals, metalloid (semi-metals) and non-metals. Most of the non-metals are molecular materials while most of the metalloids have an extended network-covalent structure.
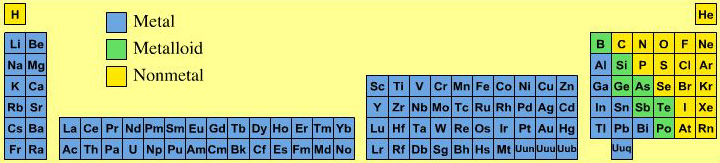
Elsewhere in the chemogenesis web book, material type is discussed in terms of the Laing Tetrahedron, an analysis that classifies binary materials in terms of four extreme types: metallic, ionic, molecular and network. However, none the chemical elements present as ionic materials, only as metals, molecular (van er Waals) and network materials:
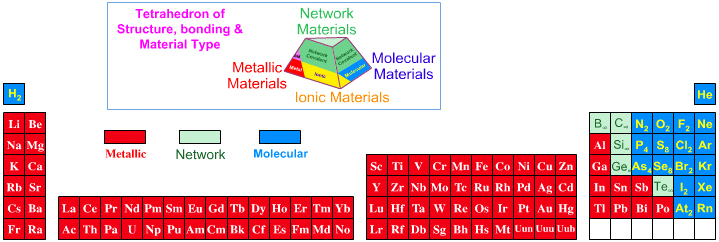
The elements B, C, Si, P, S, Ge, As, Se, Sn, Sb and Te can form allotropes: pure elemental substances that can exist with different crystalline structures from the Wikipedia. Allotropes may be metallic, network or molecular.
By Mark Leach
| Year: 2004 | PT id = 125, Type = data |
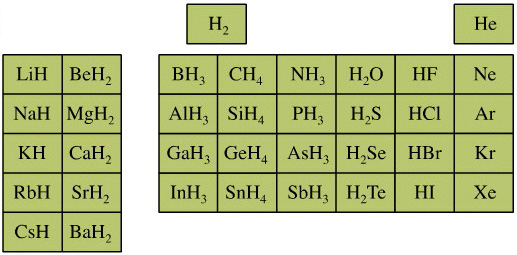
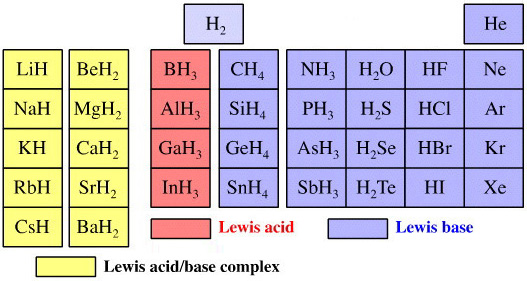
Elemental Hydride Types Periodic Table
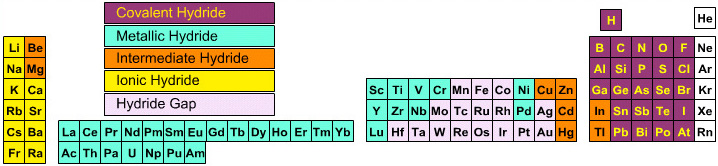
- Ionic or Salt-Like Hydrides: Molten LiH conducts electricity and hydrogen gas is liberated at the anode confirming presence of hydride ion H–. The crystal structures show an ionic lattice, and not an LiH molecular lattice.
- Covalent Hydrides are formed by the p-Bolock elements.
- Metallic or Interstitial Hydrides are formed by many d-block and f-block elements when heated with hydrogen under pressure. The hydrides tend to be non-stoichiometric and they may be of variable composition.
- There is a Hydride Gap where elements do not form hydrides. This roughly maps to the Siderophile Elements of the geologist's periodic table (below).
- The Intermediate Hydrides do not fit: beryllium hydride is polymeric, (BeH2)n. Others have properties between metallic and covalent.
The main group elemental hydrides are all well known reagent chemicals. The main group hydrides always give the lowest and most common oxidation state, and all chemicals are molecular in the gas phase. The Group I and II hydrides are ionic materials, but they can be vaporised to give the molecular form.
The chemicals present and behave as Lewis acids, Lewis bases or Lewis acid/base complexes, here:
By Mark Leach
| Year: 2004 | PT id = 126, Type = data |
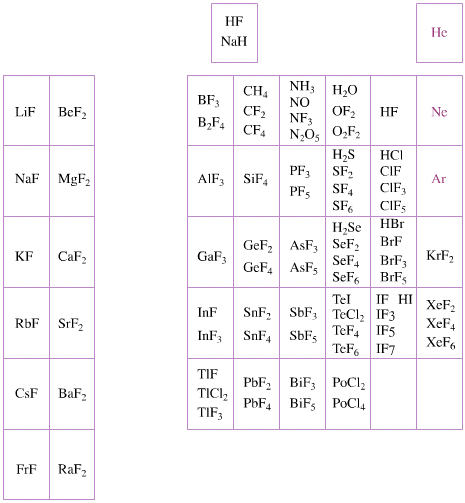
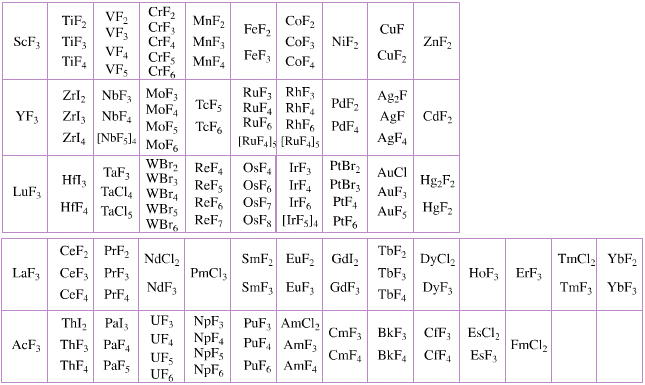
Elemental Oxidation States Periodic Table
The periodic table of fluorides (mainly) shows the range of possible oxidation states. Note that lithium, by way of example, is deemed to have two oxidation states: Li0 (the metal), and Li+ (the lithium ion):
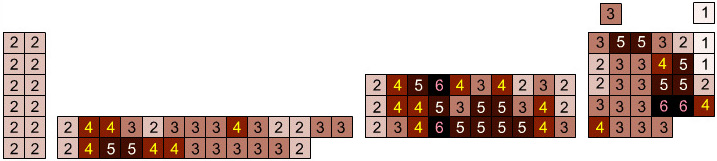
There are a few exceptions and points to note:
- There is a general increase in the number of possible oxidation states towards the lower right hand side of the periodic table.
- Nitrogen(V) fluoride, NF5, is not known, but the nitrogen(V) oxide is: N2O5.
- PtBr2 and PtBr3 are known, but PtF2 and PtF3 are not.
- All elements are known in the zero oxidation state, but apart from: He, Ne & Ar, and these are not shown in the diagram below.
- All data is from WebElements.
By Mark Leach
| Year: 2004 | PT id = 132, Type = data |
Mass Anomaly Periodic Table
Pairs of atoms where atomic mass does not follow atomic number.
|
Co
|
=
|
58.933 |
Ni
|
=
|
58.69 | ||
|
Ar
|
=
|
39.948 |
K
|
=
|
39.098 | ||
|
Te
|
=
|
127.60 |
I
|
=
|
126.90 |

Nature's little quirk – due to the intricacies of nuclear chemistry and isotopic abundance – caused no end of difficulties to the developers of the periodic table in the mid-nineteenth century. Scientists could determine atomic mass, but knew nothing of protons or atomic numbers.
The tellurium-iodine anomaly was a particular problem.
By Mark Leach
| Year: 2004 | PT id = 135, Type = data |
Phase State: Solid, Liquid, Gas at 20°C & 700°C
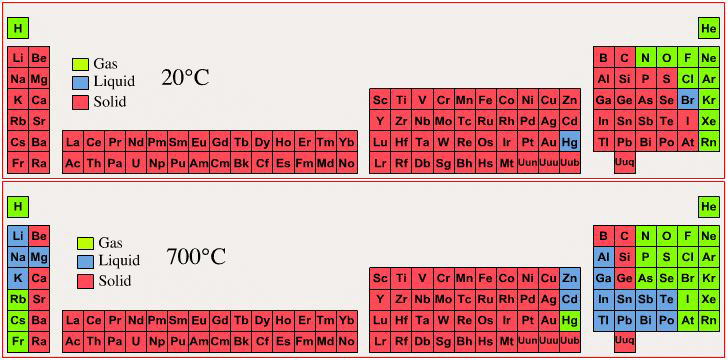
By Mark Leach
| Year: 2004 | PT id = 136, Type = data |
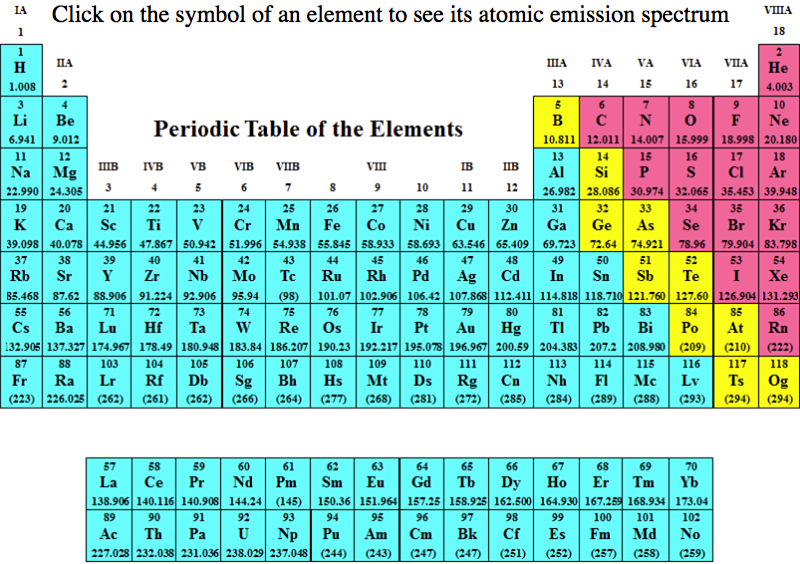
Atomic Emission Spectra Periodic Table
Department of Chemistry at PennState has a dynamic periodic table, here, which shows the atomic emission spectra of the elements:
| Year: 2004 | PT id = 139, Type = data |
Organic Chemist's Periodic Table
Organic chemistry is dominated by carbon, hydrogen, oxygen and nitrogen. Other elements are commonly encountered in the organic lab, others less commonly and some... almost never at all...
A less than useful formulation (!):

followed by a slightly more useful organic chemist's periodic table:
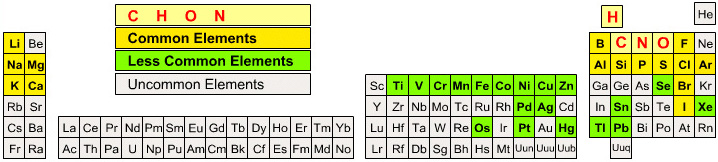
By Mark Leach
| Year: 2004 | PT id = 140, Type = data |
Inorganic Chemist's Periodic Table
Every element has a specialist, somewhere, for whom it is the most important element.

By Mark Leach
| Year: 2004 | PT id = 143, Type = data misc |
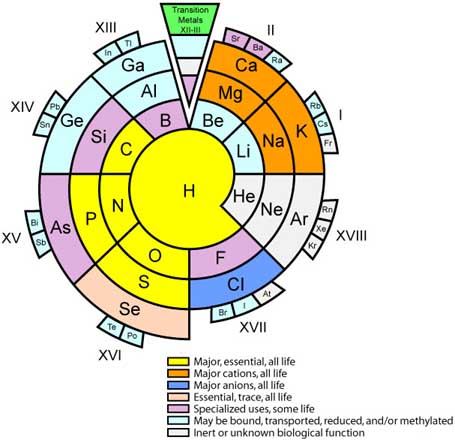
Biologist's Periodic Tables
A periodic table showing where biologically essential (green), essential trace (purple), toxic (red), radioactive (yellow) and of low – but not zero– biological impact (gray) elements are found. Only highly toxic elements are shown in red. Li (as Li+) is biologically active and is used as an antidepressant.
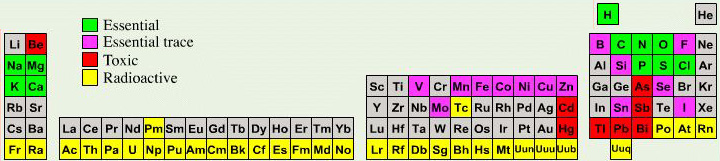
By Mark Leach
or here:
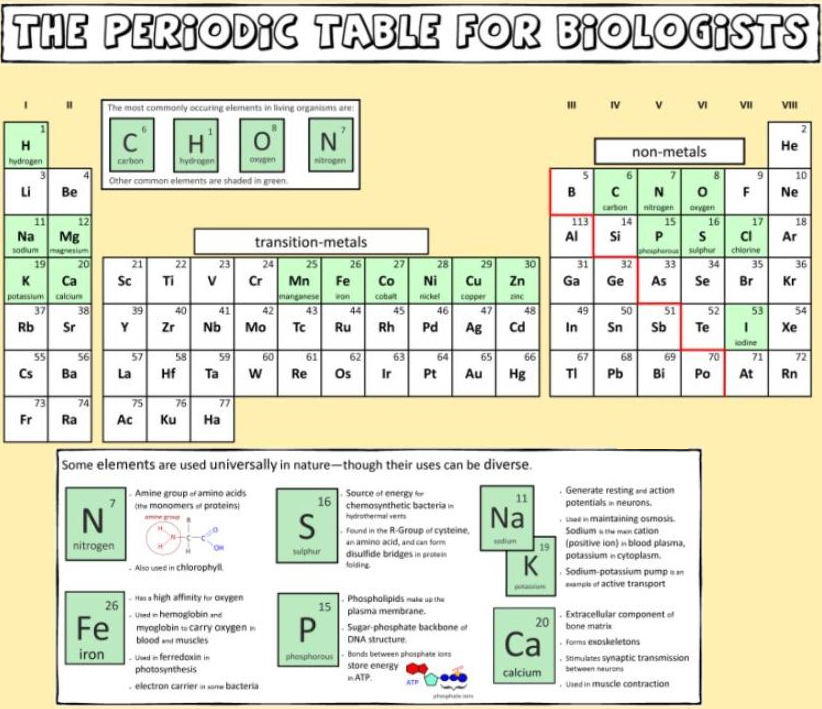
And a periodic table for biologists from Science Videos:
| Year: 2004 | PT id = 177, Type = non-chem |
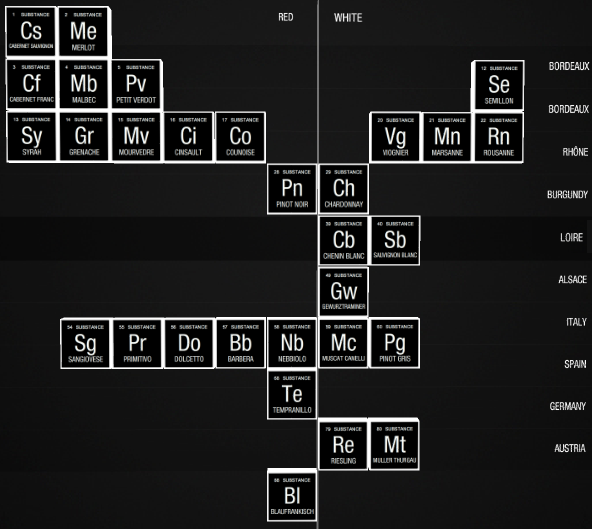
Wine
A Wasington State periodic table of wine types from Wines of Substance:
| Year: 2004 | PT id = 181, Type = non-chem |
Printmaking
The periodic table Printmaking Project:
| Year: 2004 | PT id = 256, Type = non-chem |
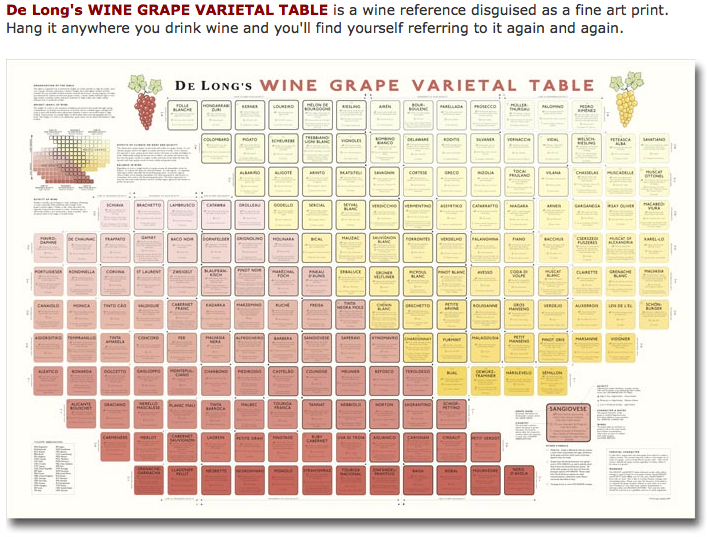
De Long's Wine Grape Varietal Table
| Year: 2004 | PT id = 271, Type = non-chem |
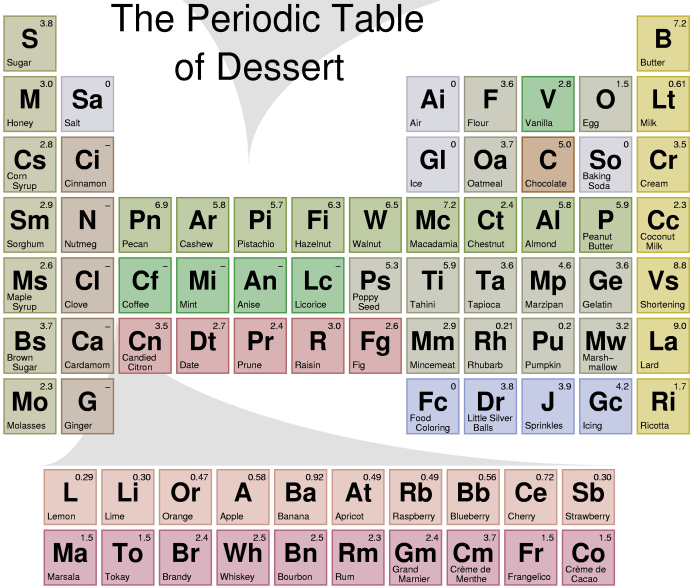
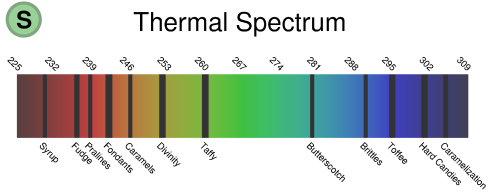
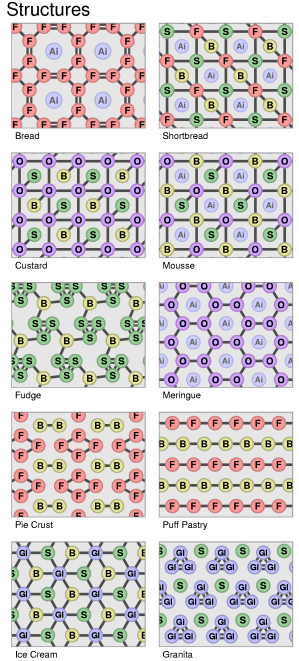
Dessert Periodic Table
A Scientific and Rigorous approach to patisserie -- in Full Color by Andrew Plotkin (2003)
You've seen those charts that say, like, "Periodic Table of the Vegetables" or "Periodic Table of the Sausages"? They annoy me because they are not periodic. They have no vertical or horizontal correspondences. The actual periodic table of chemical elements has structure -- that's why it's cool. Thus, my contribution to the field:
| Year: 2004 | PT id = 406, Type = non-chem |
Blues Periodic Table
A Periodic Table of The Blues by Instruments For Research and Industry:

| Year: 2004 | PT id = 607, Type = formulation |
Piano Periodic Table
A Piano Periodic Table from Claude Bayeh:


| Year: 2004 | PT id = 968, Type = data misc |
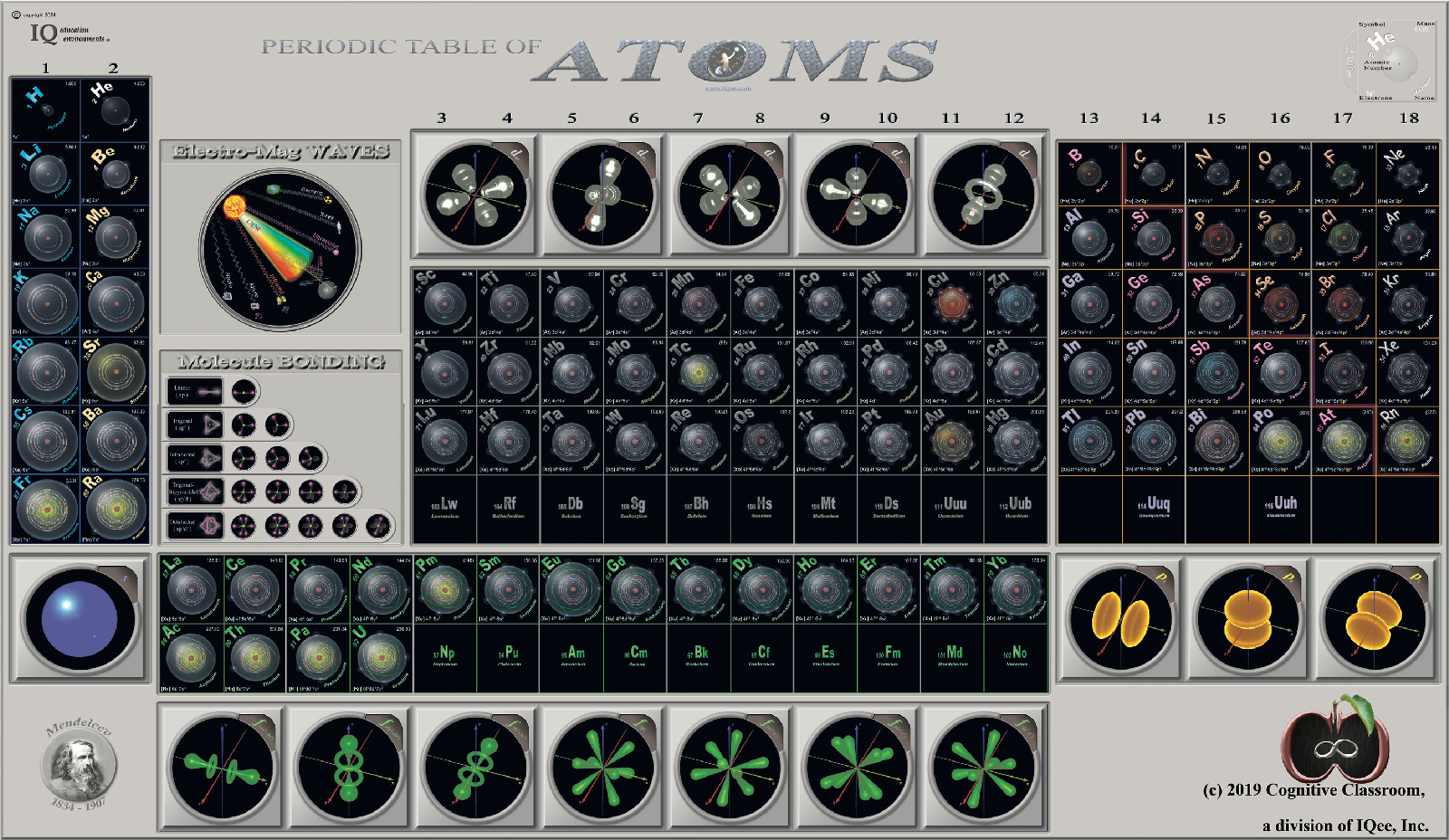
Cognitive Classroom's Periodic Table of Atoms
From Cognitive Classroom, a Periodic Table of Atoms. Richard Lambrecht writes:
"We have developed a visual periodic table that groups by orbitals, making He no longer contentious. But by including an orbital cloud, we give the student a great offset to the Bohr model used to place each and every single electron in the periodic table."
Click image or here to enlarge:
| Year: 2004 | PT id = 1096, Type = misc |
Classroom Kids Periodic Table
From a paper by René Vernon, a drawing of the elements as classroom personality kids, drawing by Richard Thompson 1957-2016.
From a National Geographic coffee table book: Curt Suplee, The New Everyday Science Explained, National Geographic Society, Washington DC, p. 130 (2004). The undated credit is given to Richard Thompson.
- Sodium is actively practicing a "salt-formation" passing play with corrosive chlorine, who devours the delivery.
- Potassium, magnesium and barium are having an active metal huddle.
- The two hydrogens are hot desking.
- Iron is reading a weightlifting workout book.
- Platinum introduces gold to iridium, all three being noble metals.
- Lead, as a heavy frontier metal, is playing air guitar.
- Carbon is having a link up with frontier metal silver (masquerading as a transition metal); an unidentified frontier metal; and two "other" nonmetals.
- Silicon, as a metalloid and a semiconductor, is catching up on an assignment.
- Helium and neon, having each had a cup of chamomile tea during the break, are sleeping.

| Year: 2004 | PT id = 1100, Type = formulation review |
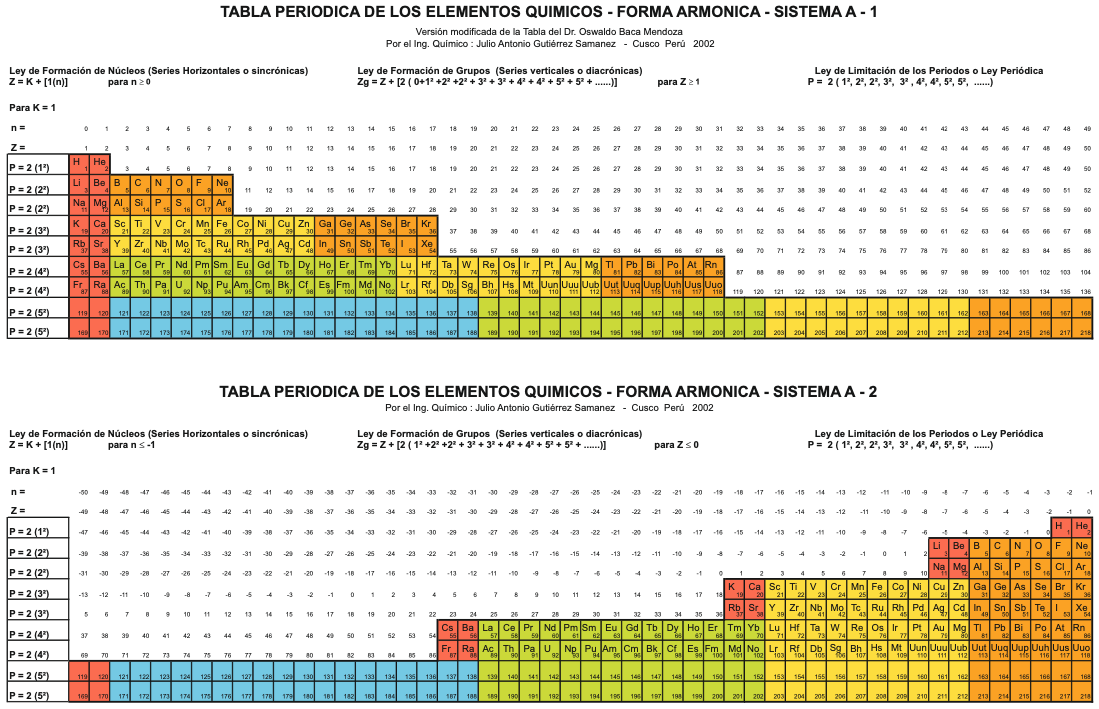
Sistema Periódico Armonico de Gutierrez-Samanez
A digitised 2004 book by book by Julio Gutiérrez-Samanez.
Julio writes:
"These matrix tables are inspired by the method used by the Peruvian chemist Oswaldo Baca Mendoza (1908-1962).
"The tables are read in this way: The Law of Formation of nuclei generates all the horizontal series Z, is dependent on n (series of integers numbers) and a constant K = 1. In the step-to-right tables (n) it will be equal to or greater than 0. In Janet's Left-Step table, (n) will be less than or equal to (-1). The values of this series Z will serve as a constant for the second law.
"The Group Formation Law or vertical series. Its generate the numeric values of the columns either from left to right or from right to left. In system A -1: With the first law: n = 0, then Z = 1. The vertical series Zg = 1, 3 11, 19, 37, 55, 87 ... That is: 1H, 3Li, 11Na, 19K, 37Rb, 55Cs, 87Fr, 119, 169 ... Changing the values of n or Z, all the columns of the table will be obtained.
"In system A -2: With the first law: on the left, n = -1, then Z = 0. The vertical series Zg will be: 2He, 10Ne, 18Ar, 36Kr, 54Xe, 86Rn, 118Og, 168, 218. .. Similarly, changing the n or Z values, we can fills the columns of the table. In system B -1: With the first law: for n = 0, then Z = 1. The vertical series Zg will be; 1H, 3Li, 5B, 13Al, 21Sc, 39Y, 57La, 89Ac, 121, 171 ... By varying the values of n or Z, the entire table is filled. In system B -2: (Its mathematizes the Janet system). With the first law: on the left, for n = -1, then Z = 0.
"The vertical series Zg will be; 2He, 4Be, 12Mg, 20Ca, 38Sr, 56Ba, 88Ra, 120, 170, 220 ... By varying the values of n or Z, the entire table is filled. The third law of the limiting the periods or periodic law, appears graphically, by comparison between rows: For example: in table B -1, in column Z = 3, after 1H and 2He, en of the first horizontal line, the value 3 appears, which is already entered in the first column as 3Li, therefore, that part of the first horizontal row (from 3 to 50) is deleted.
"The same happens with the number 5 in column 3, which is already in the first column as 5B, therefore it will be deleted in the second row from 5 to 52. The same applies to pair 13, 21 of the column Z = 9, same, with the pair 39, 57 of the column Z = 19 and of the pair 89, 121 of the column Z = 33. For that reason the periods: P are duplicated function: 2 (1 ^ 2), 2 (1 ^ 2), 2 (2 ^ 2), 2 (2 ^ 2), 2 (3 ^ 2), 2 (3 ^ 2) .... = 2, 2, 8, 8, 18, 18, 32, 32 ... and the forms are exact and staggered. The colors represent the quantum functions: s (red), p (orange), d (yellow), f (green), g (blue)."
 |
 |
 |
| What is the Periodic Table Showing? | Periodicity |
© Mark R. Leach Ph.D. 1999 –
Queries, Suggestions, Bugs, Errors, Typos...
If you have any:
Queries
Comments
Suggestions
Suggestions for links
Bug, typo or grammatical error reports about this page,please contact Mark R. Leach, the author, using mark@meta-synthesis.com
This free, open access web book is an ongoing project and your input is appreciated.