Periodic Table |
 |
 |
 |
 |
 |
 |
 |
| What is the Periodic Table Showing? | Periodicity |
The INTERNET Database of Periodic Tables
There are thousands of periodic tables in web space, but this is the only comprehensive database of periodic tables & periodic system formulations. If you know of an interesting periodic table that is missing, please contact the database curator: Mark R. Leach Ph.D. The database holds information on periodic tables, the discovery of the elements, the elucidation of atomic weights and the discovery of atomic structure (and much, much more).
The 10 most recent entries to the database:
| Year: 2026 | PT id = 1397, Type = formulation review 3D |
Spin and The Forth Dimension
A paper (click to view pdf) by V.V. Varlamov of Siberian State Industrial University, Novokuznetsk, Russia who writes:
"A group-theoretic interpretation of the periodic system of elements is given within the framework of the weight diagram of the Lie algebra SO(4,4) of the fourth rank, where the four quantum numbers n, l, m, scorrespond to the eigenvalues (weights) of the Cartan generators of the maximal Abelian subalgebra (the maximal torus of the group SO(4,4)). Etc."
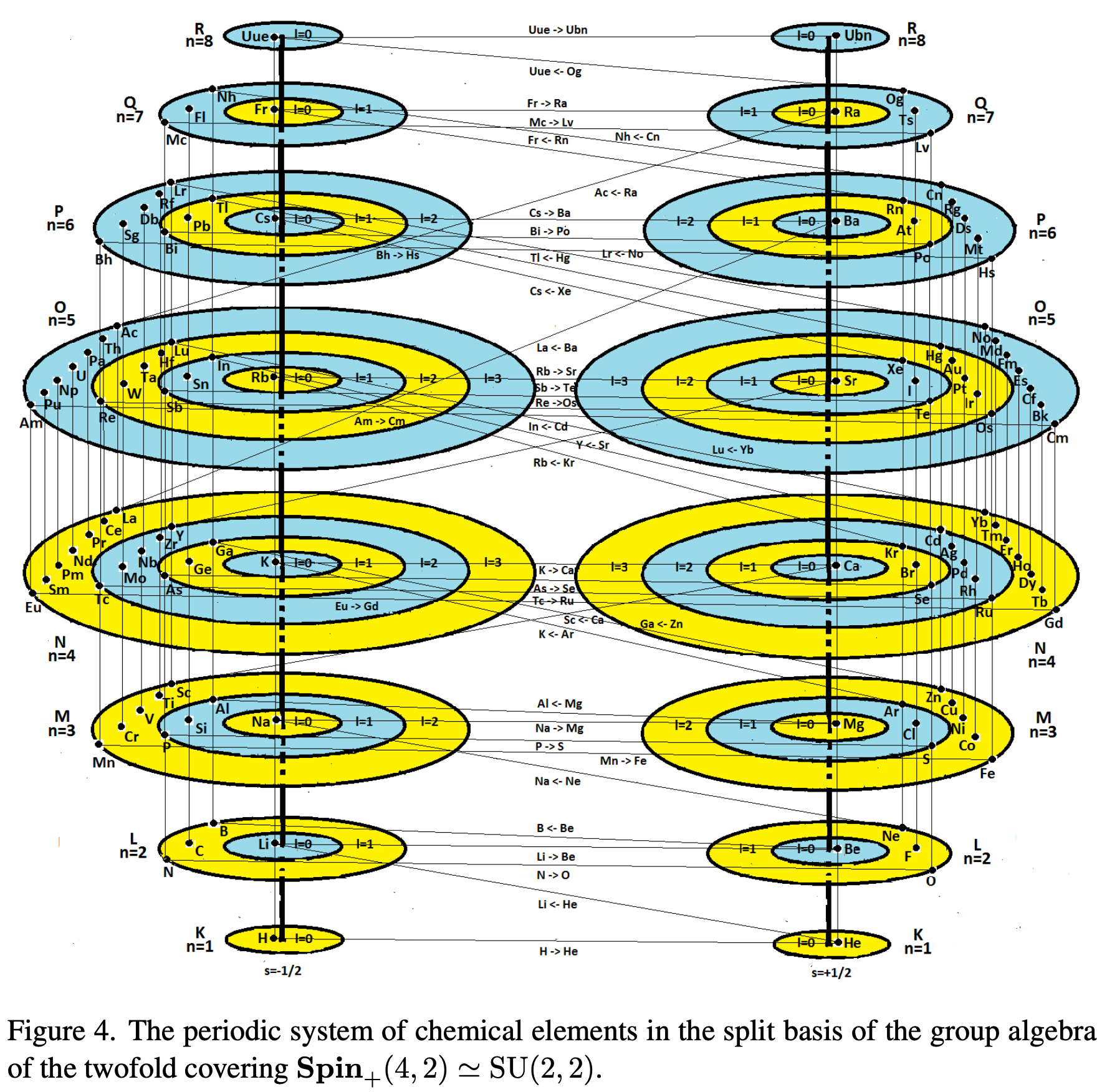
The paper generates a Janet Left-Step formulation:
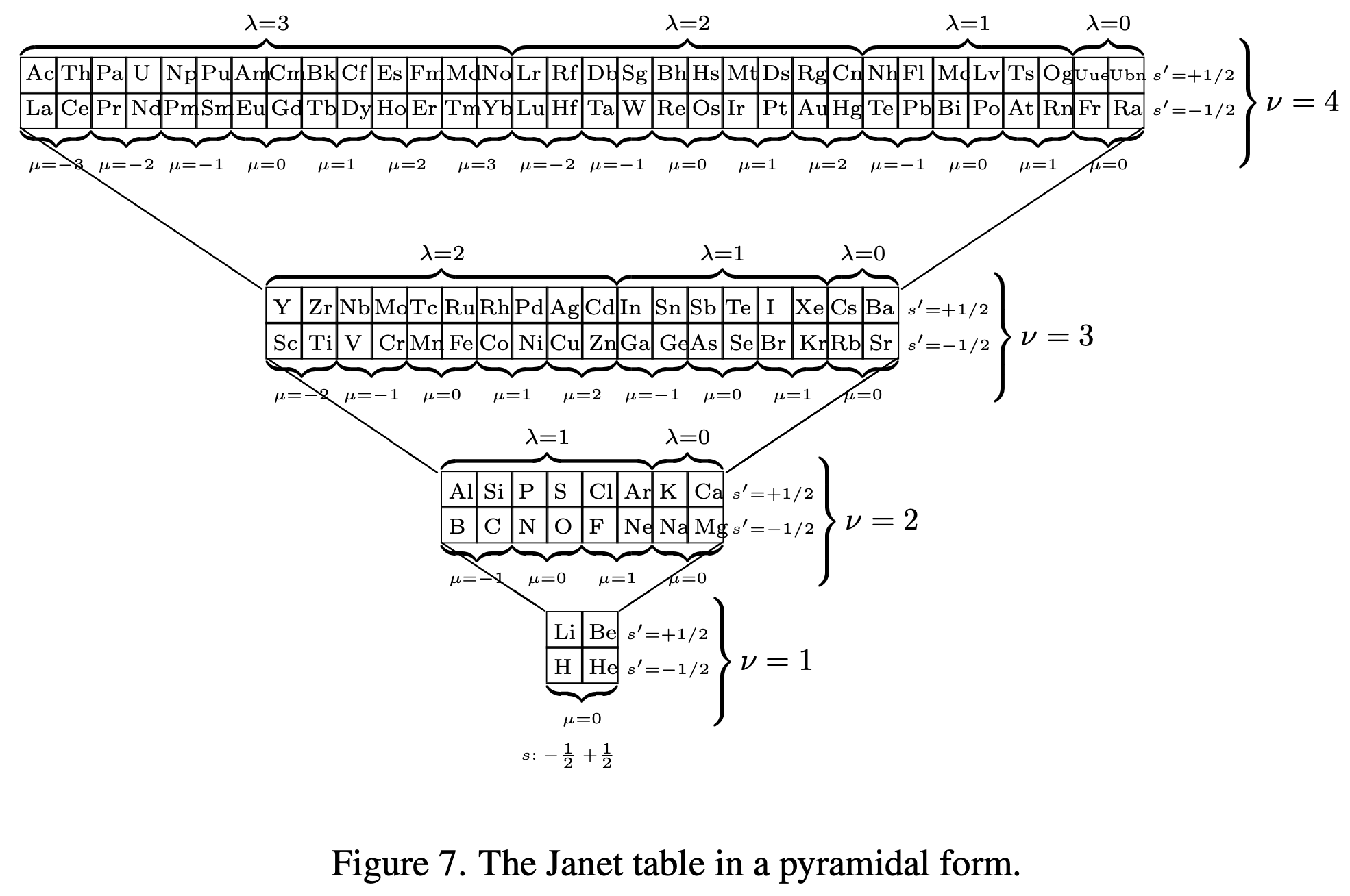
Thanks to Eric Scerri for the tip!
| Year: 2026 | PT id = 1396, Type = element structure review |
Quest To Understand Where Atoms End
By Philip Ball in Chemistry World, who writes:
"There is no consensus. 'We have some models and theories [of atomic size] but none have been really amenable to experimental verification,' says Amin Alibakhshi at the Technical University of Dortmund in Germany. 'That’s why we have many different definitions, like van der Waals radii, covalent radii, and so on.' But is each of these definitions in the end a rather arbitrary attempt to carve up the smooth and continuous electron density so that it seems to have hard edges? Or might there be, after all, some deeper and more objective meaning to the size of an atom?"
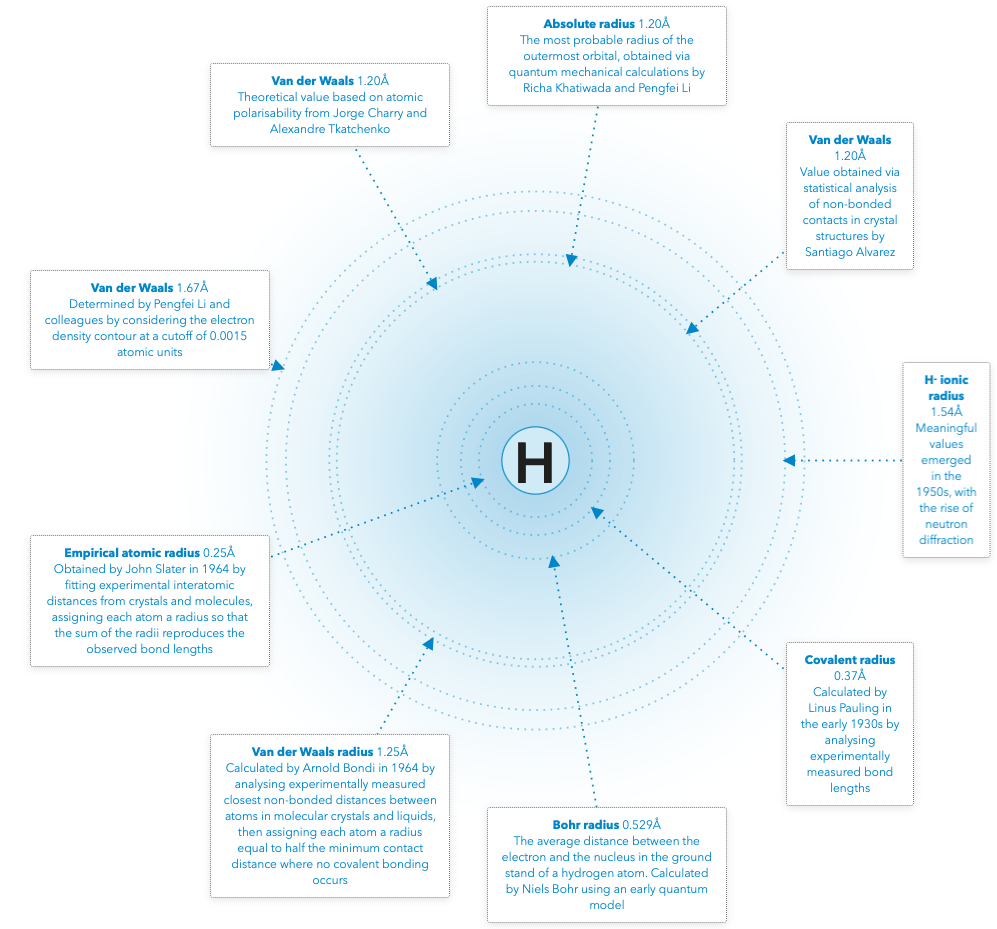
| Year: 1995 | PT id = 1395, Type = misc |
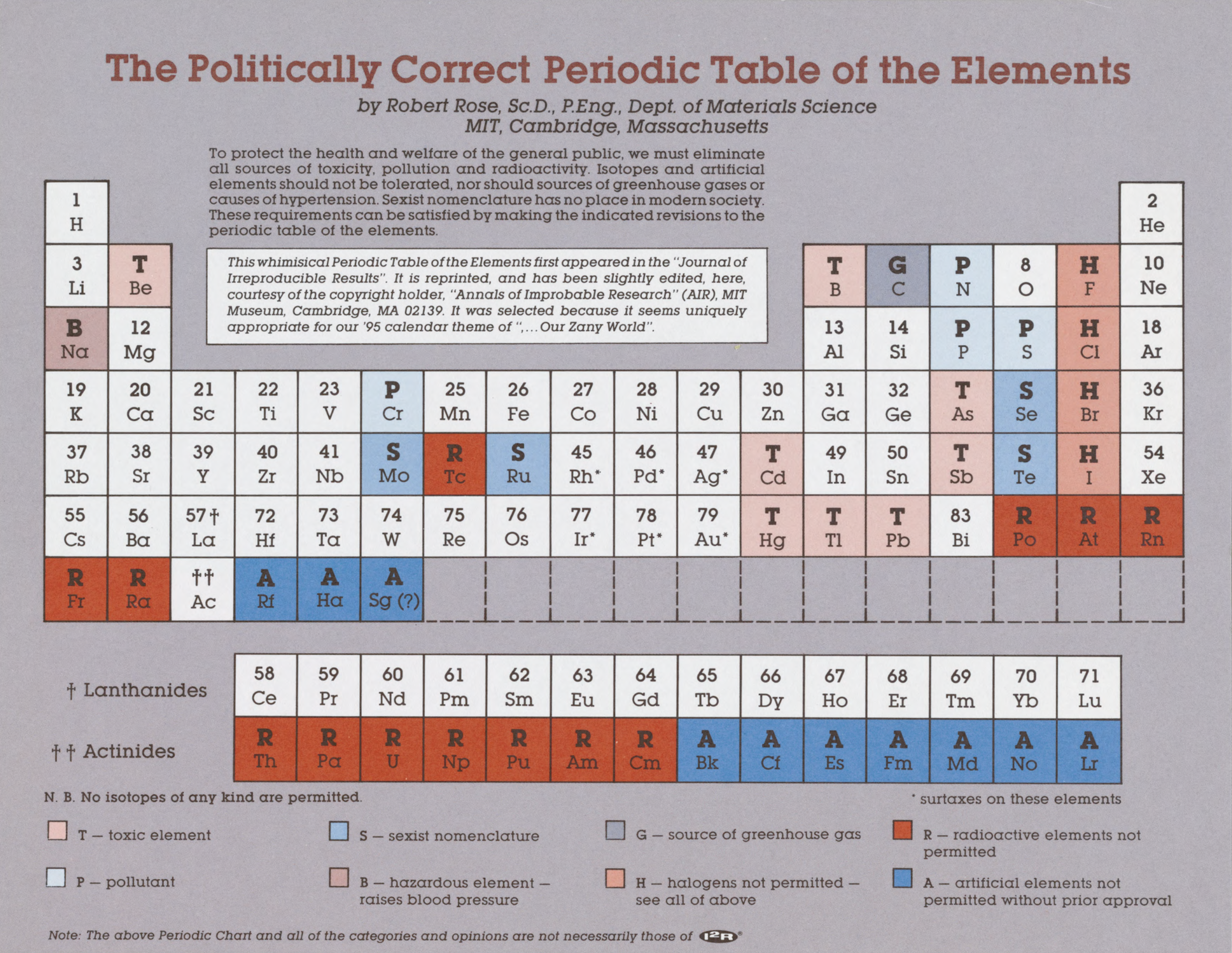
Politically Correct Periodic Table of the Elements
By Robert Rose of MIT, appearing in the I2R calendar. (We guess the date at 1995.)
From Craigslist:
"Instruments for Research and Industry, better known to scientists as I2R (eye-squared-are), was founded in the 1960s. Soon after, they started offering humorous, science and research themed calendars as part-catalog, part-advertising, part-promotional and completely entertaining material. I2R was a 'Profits for Peace' company. I2R donated a portion of their profits to charitable causes. (The time was the 1960s - 1970s, the Vietnam War era. I2R estimated how much of their business taxes were going to the Department of Defense and then they offset that with donations to charitable causes.)"
Text:
"To protect the health and weltare of the general public, we must eliminate all sources of toxicity, pollution and radloactivity. Isotopes and artificial elements should not be tolerated, nor should sources of greenhouse gases or causes of hypertension. Sexist nomenclature has no place in modern society. These requirements can be satisfied by making the indicated revisions to the periodic table of the elements.
"This whimisical Periodic Table of the Elements first appeared in the Journal of Irreproducible Results. It is reprinted (slightly edited) here."
The PC-PT identifies:
- T: Toxic Elenents
- S: Sexist Nomenclature
- G: Greenhouse Gas Source
- R: Radioactive Elements (not permitted)
- H: Hazardous Element (raises blood pressure)
- H: Halogens (not permitted)
- A: Artificial Elements (not permitted without prior approval)
Thanks to Eric Scerri for the tip!
| Year: 1935 | PT id = 1394, Type = formulation structure element |
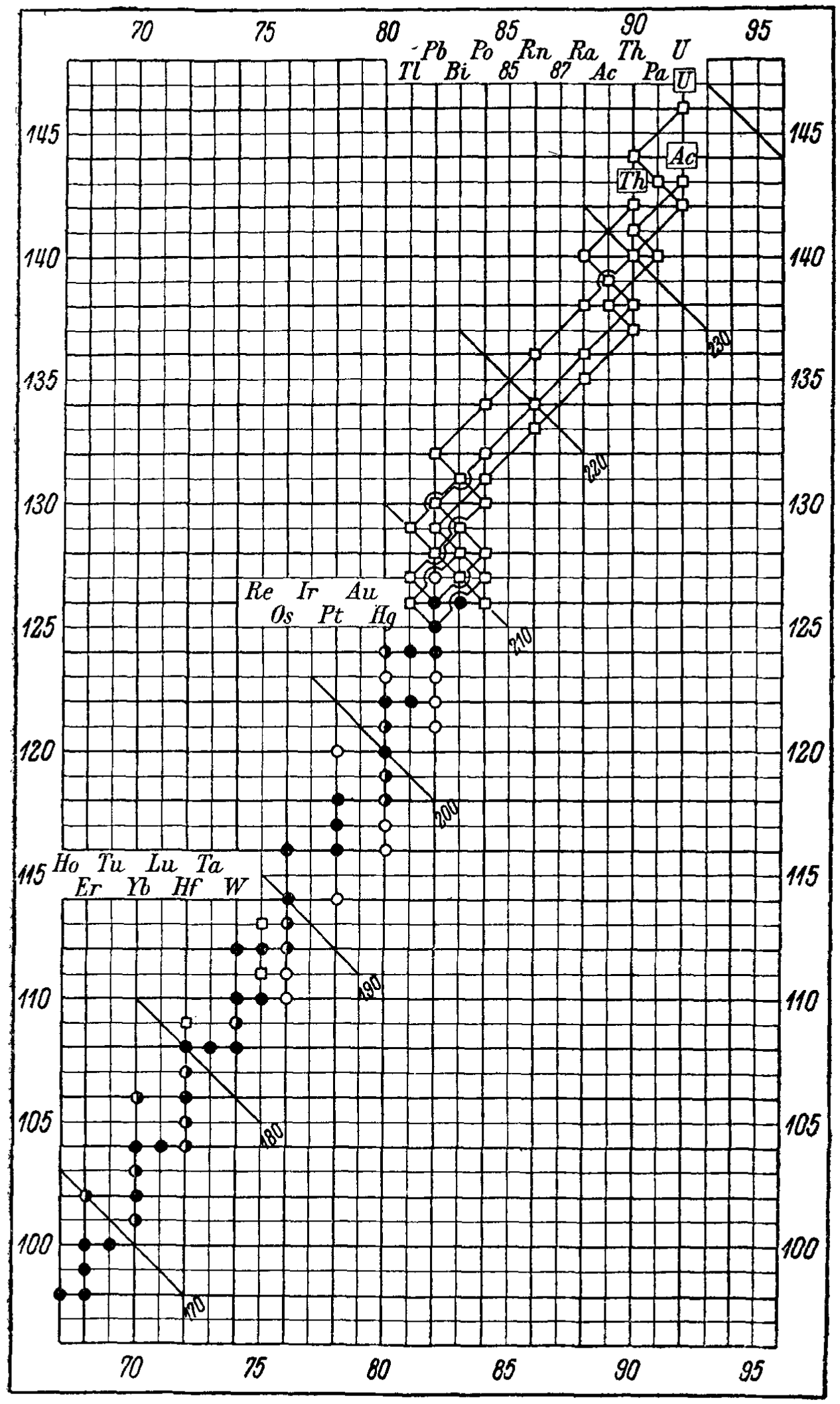
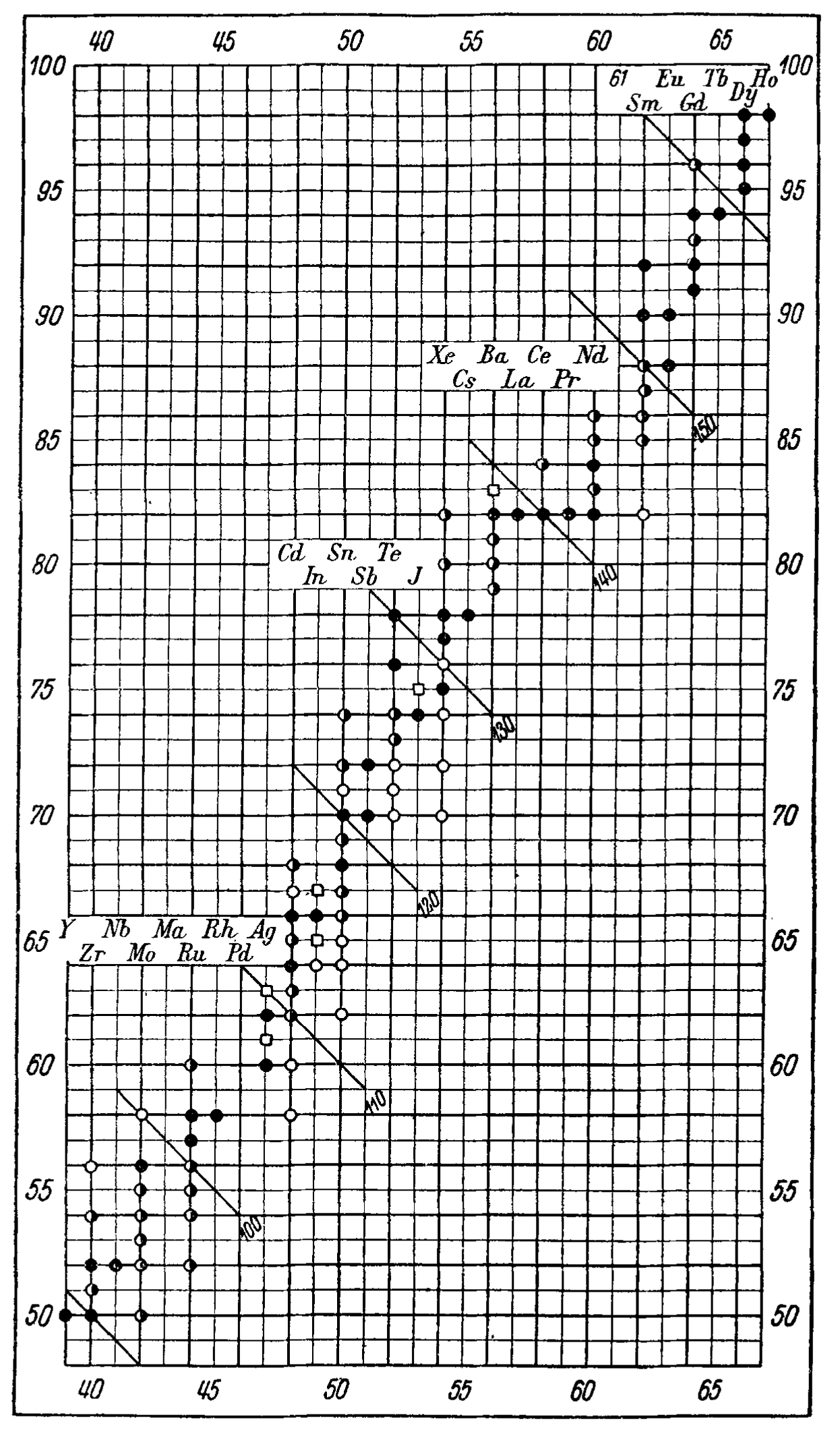
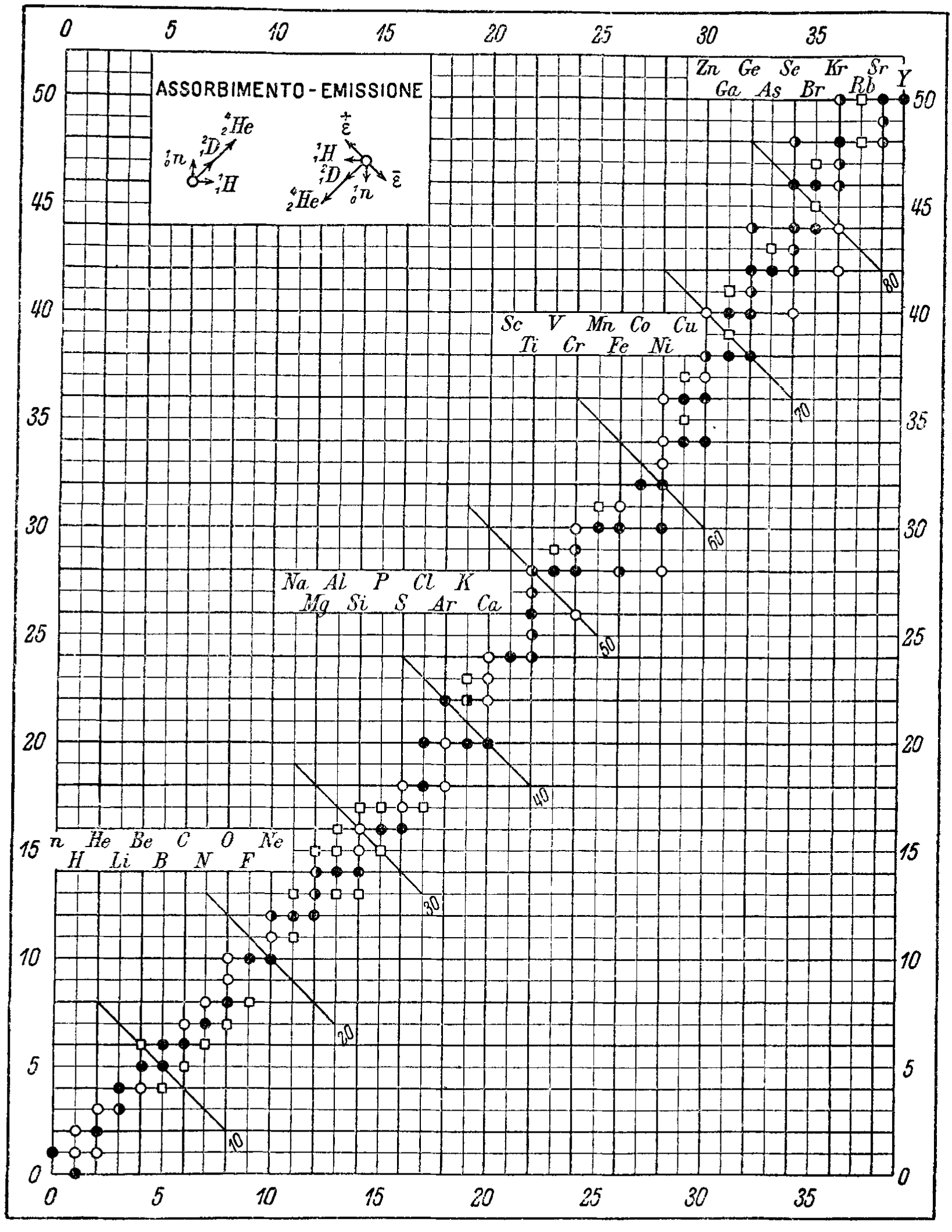
Tabelle Riassuntive E Bibliografia Delle Trasmutazioni Artificiali (Summary Tables And Bibliography Of Artificial Transmutations)
A 1935 paper by Fae which predates the Segrè Chart by 10 years: Fea, G. Tabelle Riassuntive e Bibliografia Delle Trasmutazioni Artificiali. Nuovo Cim 12, 368–406 (1935). https://doi.org/10.1007/BF02958685 (Thanks to Mario Rodríguez Peña who found this paper!)
Abstract (Google Translate):
"Given the significant developments in the study of artificial transmutations, especially after the impetus provided by the discovery of induced radioactivity, it seemed useful to the writer to summarize in some synoptic tables what has been obtained to date by the many researchers who have studied the topic.
"An extensive bibliography of the works consulted for the compilation of the tables follows, as well as a table representing, in the neutron-proton diagram, what is known about stable and radioactive isotopes."
| Year: 2026 | PT id = 1393, Type = element review |
What is an element, and how is it defined in the IUPAC Gold Book?
A recent publication by Eric Scerri: What is an element, and how is it defined in the IUPAC Gold Book?
Read the paper or download the PDF here, or go to page 36 of the Chemistry International journal here.

| Year: 1930 | PT id = 1392, Type = formulation |
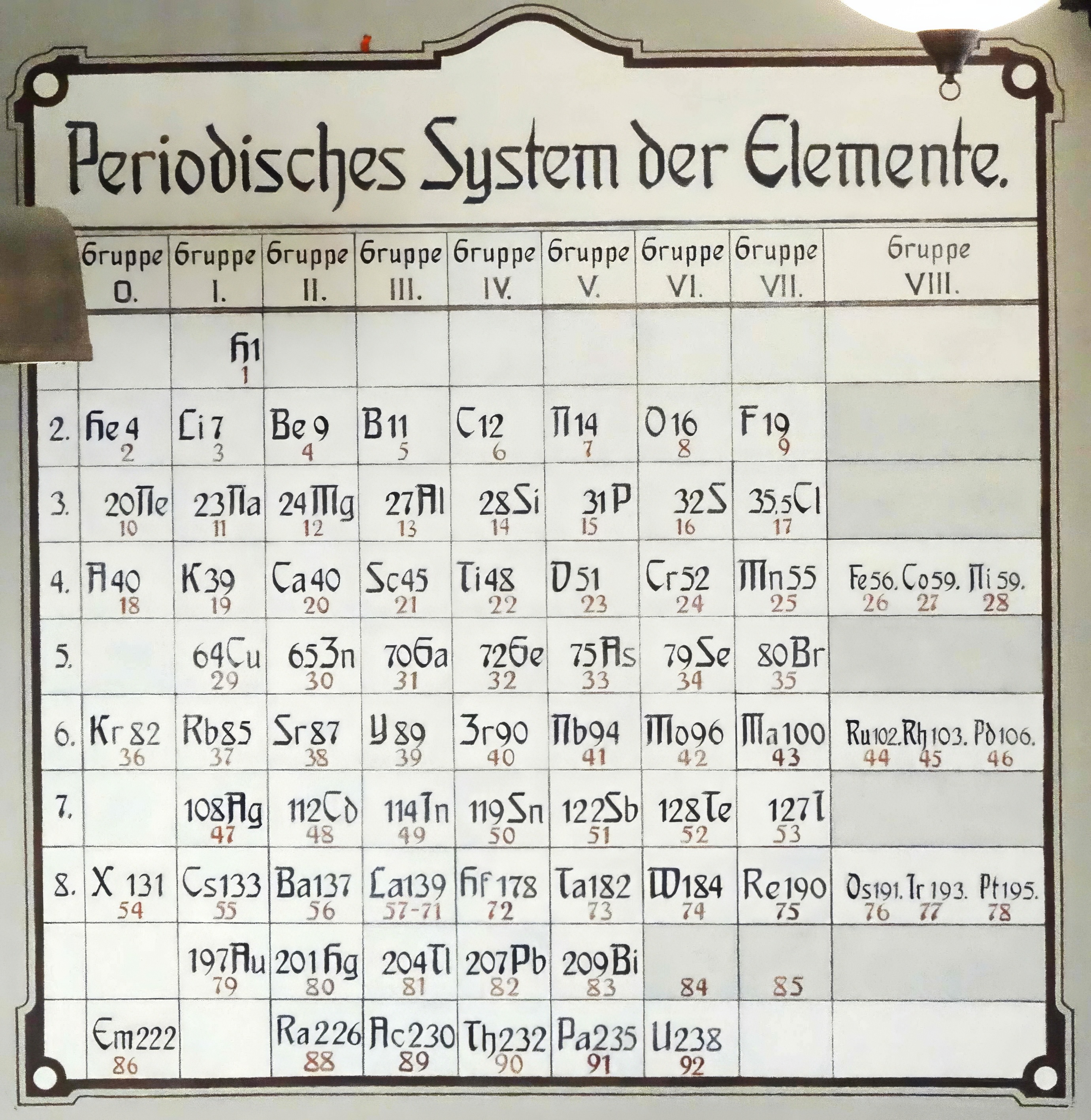
Periodisches System der Elemente
Posted on Wikimedia by LukaszKatlewa who writes:
"Periodisches System der Elemente (1904-1945, now at the Gdansk University of Technology): lack of elements: 84 polonium Po (though discovered as early as in 1898 by Maria Sklodowska-Curie), 85 astatine At (1940, in Berkeley), 87 francium Fr (1939, in France), 93 neptunium Np (1940, in Berkeley) and other actinides and lanthanides. Old symbols for: 18 argon Ar (here: A), 43 technetium Tc (Ma, masurium, 1925, dismissed as an error and finally confirmed in 1937, Palermo), 54 xenon Xe (X), 86 radon, Rn (Em, emanation)."
Mark Leach writes: "I would guess the date at 1930."
| Year: 2026 | PT id = 1391, Type = element review |
6 Times Scientists Were Wrong About the Periodic Table
A video from SciShow discussing six times scientists made incorrect predictions about chemical elements: Coronium; Nebulium (Nebulium, Nephium, Nephelium); Masurium (Davium, Leucium, Neponium... actually Technecium); Florencium/Illinium (actually Promethium); Ausenium; and Hesperium.
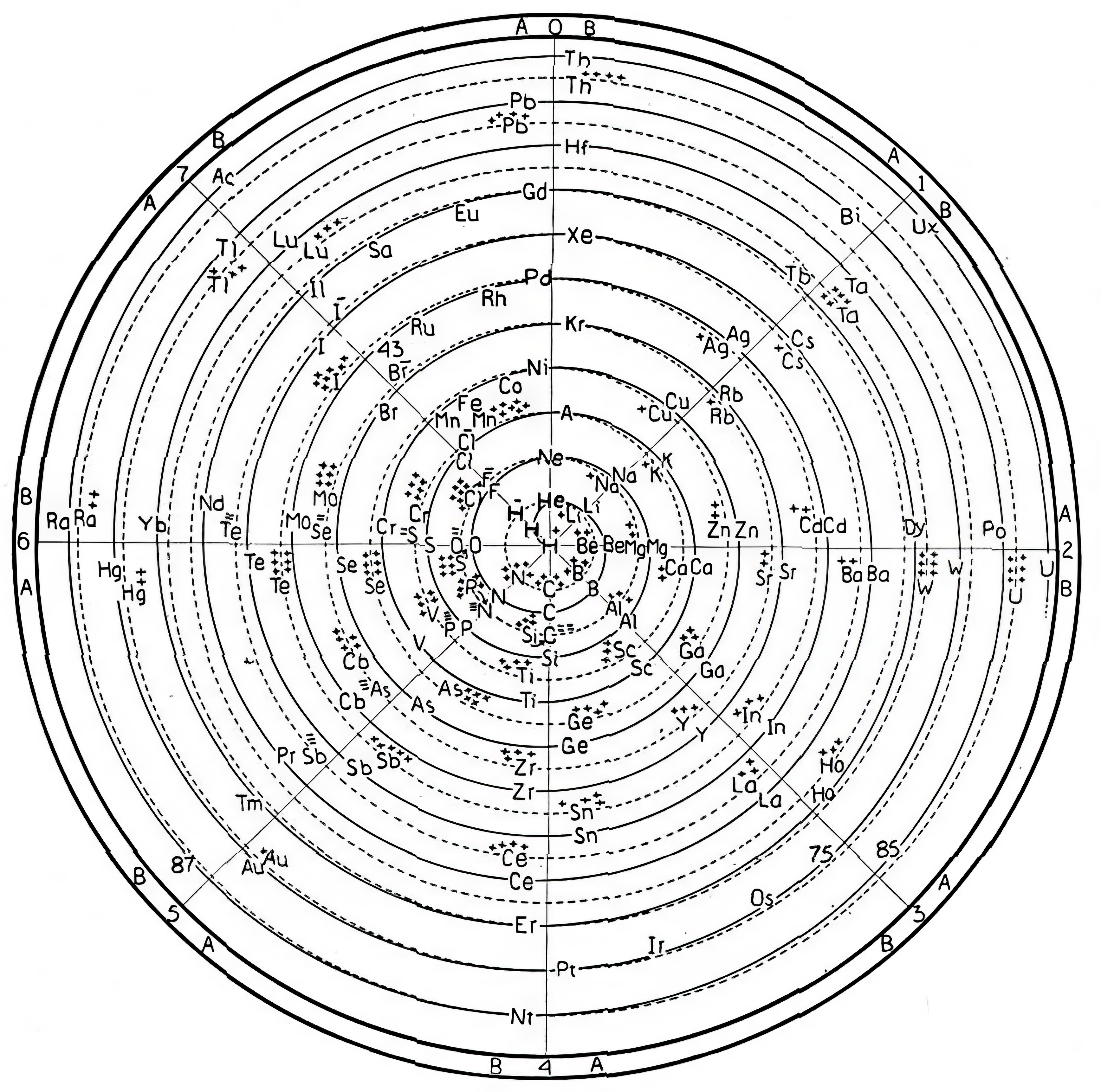
| Year: 1928 | PT id = 1390, Type = formulation spiral |
Another Attempt to Base a Classification of The Elements on Atomic Structure
Stewart, O.J., Another Attempt to Base a Classification of The Elements on Atomic Structure, J. Chem. Educ. 1928, 5, 1, 57 doi.org/10.1021/ed005p57:
| Year: 2023 | PT id = 1389, Type = review formulation |
La Tabla Periódica. El poder de la sistematización. La importancia de la precisión
A video of a presentation (in Spanish) by Manuel Yáñez, a professor at the Autonomous University entitled: La Tabla Periódica. El poder de la sistematización. La importancia de la precisión (The Periodic Table. The power of systematization. The importance of precision.)
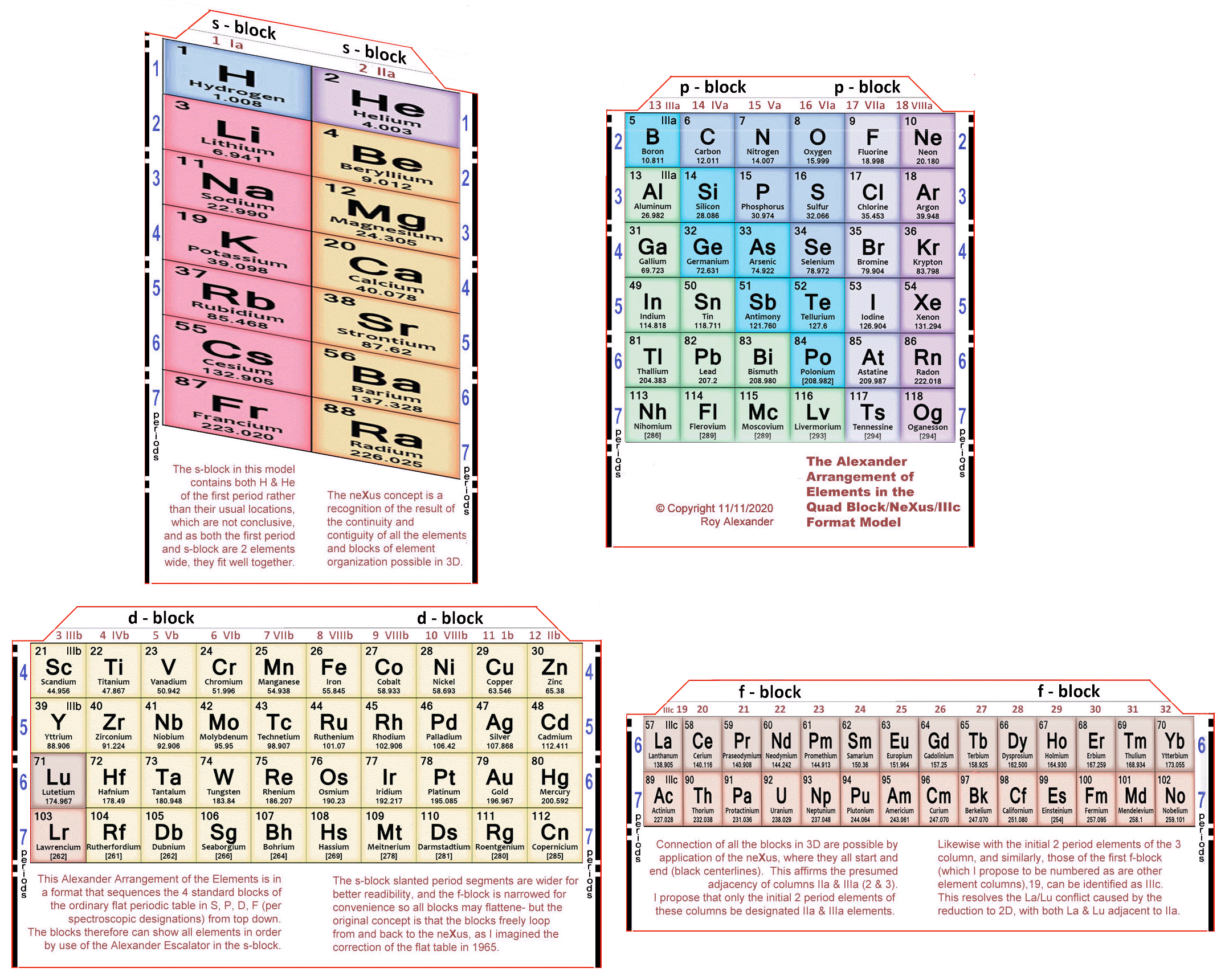
| Year: 2020 | PT id = 1388, Type = formulation 3D spiral |
Alexander's Quad Block/neXus/IIIc Model
Roy Alexander's Quad Block/neXus/IIIc Model.

 |
 |
 |
| What is the Periodic Table Showing? | Periodicity |
© Mark R. Leach Ph.D. 1999 –
Queries, Suggestions, Bugs, Errors, Typos...
If you have any:
Queries
Comments
Suggestions
Suggestions for links
Bug, typo or grammatical error reports about this page,please contact Mark R. Leach, the author, using mark@meta-synthesis.com
This free, open access web book is an ongoing project and your input is appreciated.