Periodic Table |
 |
 |
 |
 |
 |
 |
 |
| What is the Periodic Table Showing? | Periodicity |
The INTERNET Database of Periodic Tables
There are thousands of periodic tables in web space, but this is the only comprehensive database of periodic tables & periodic system formulations. If you know of an interesting periodic table that is missing, please contact the database curator: Mark R. Leach Ph.D. The database holds information on periodic tables, the discovery of the elements, the elucidation of atomic weights and the discovery of atomic structure (and much, much more).
Periodic Tables from the year 1921:
| 1921 | Margary's Periodic Table |
| 1921 | Bury's Periodic Arrangement based on Langmuir's Theory |
| 1921 | Formánek's Periodic Table |
| 1921 | Margary's Modified Table |
| Year: 1921 | PT id = 68, Type = formulation |
Margary's Periodic Table
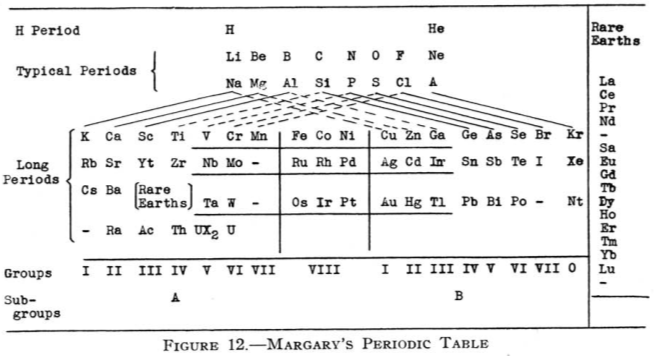
From Quam & Quam's 1934 review paper.pdf
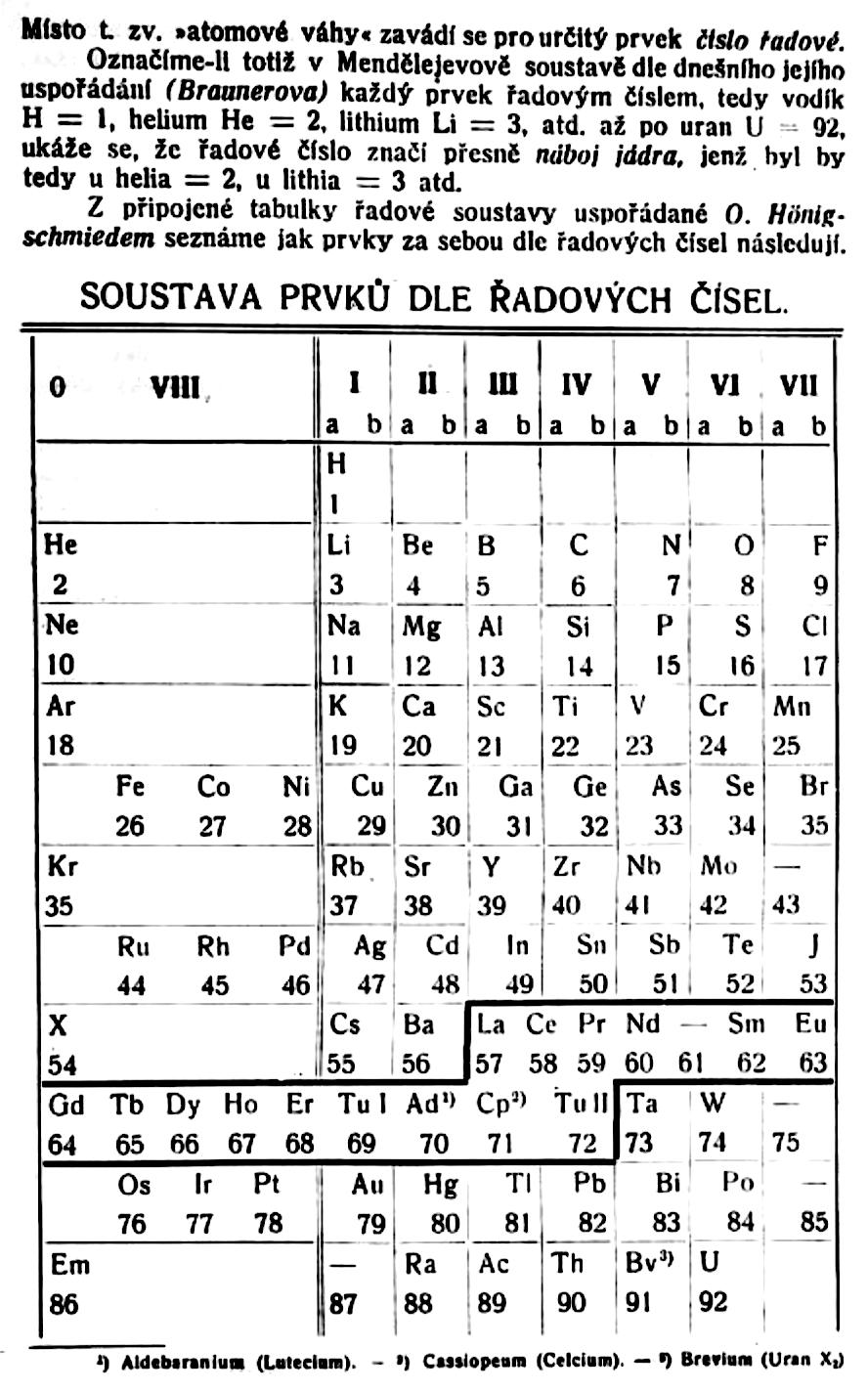
| Year: 1921 | PT id = 1020, Type = formulation |
Bury's Periodic Arrangement based on Langmuir's Theory
Using Langmuir's theory of the arrangement of electrons in atoms, J.Am.Chem.Soc., 41, 868 (1919), Charles R. Bury formulated a Periodic Arrangement: C.R. Bury, Langmuir's theory of the arrangement of electrons in atoms and molecules, J. Am. Chem. Soc., 43, 1602-1609 (1921).
This formulation seems to be the basis of Seaborg's formulations of 1939, 1942 & 1945.
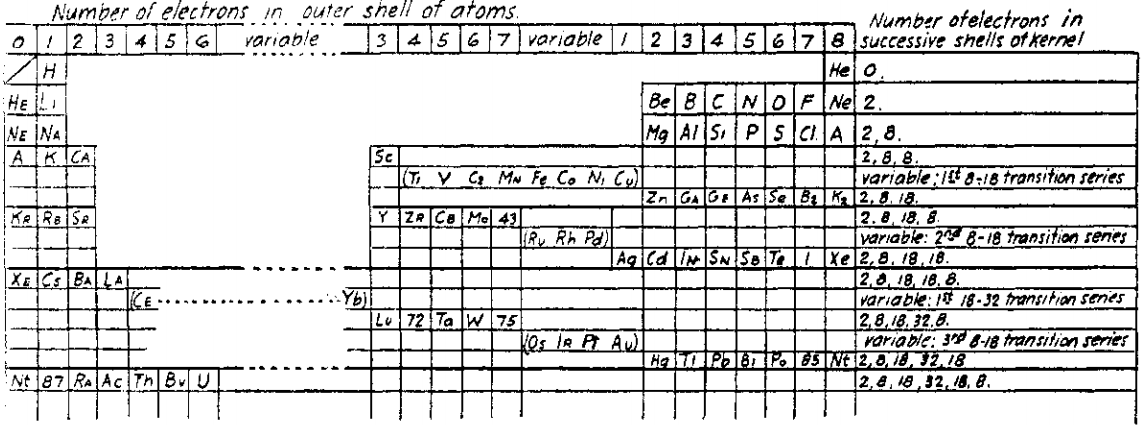
Ricardo R Contreras, Avances en Química, 14(1), 41-60 (2019), has re-drawn the Bury PT and writes [Google Translate]:
"This version emphasizes periods and electronic configurations.
"There is a long period in which the metals of titanium to copper are found, which he calls transition elements. [This formulation] leaves spaces for the element atomic number 43, technetium, discovered by Perrier Segre in 1937; for the element 72, hafnium, discovered in 1932 by D. Coster and G. von Hevesey; for the element 87, the eka-cesium, which corresponds to francium (Fr), discovered in 1939 by the French physicist Marguerite C. Perey (1909-1975) and, at the end of the group of halogens, for the element 85, the astatine (At), synthesized for the first time in 1940 by American physicists Dale R. Corson (1914-2012), Kenneth R. MacKenzie (1912-2002) and the Italian-American physicist Emilio G. Segrè (1905-1989) at the University of Berkeley (California), bombarding bismuth with particles.
"Bury uses 'A' as the symbol argon, 'Nt' (niton) for radon (Rn) and, the symbol 'Bv' (brevium) for proctactinium (Pa)."
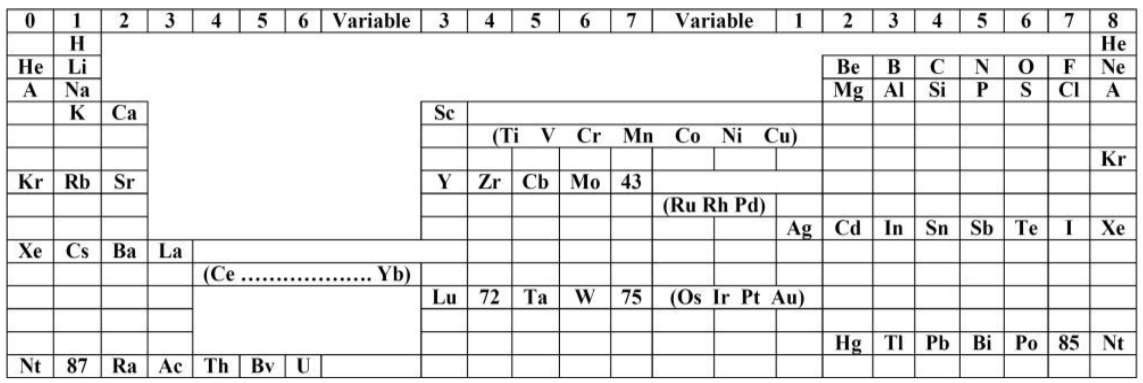
| Year: 1921 | PT id = 1192, Type = formulation |
Formánek's Periodic Table
Formánek J. 1921, Short Outline of Inorganic Chemistry (in Czech), 2nd ed., Ministerstvo zemedelstvi CSR, Praha. p. 281
René Vernon writes:
Here is an eight column table with some interesting features.
Main groups 0, Ia, IIa, Vb, VIb, and VIIb, correspond to what we have today:
- 0 Noble gases
- Ia Alkali metals
- IIa Alkaline earths
- Vb Pnictogens
- VIb Chalcogens
- VIIb Halogens
Main group IIIa is B-Al-Sc-Y... Ac whereas these days B-Al have been moved over Ga on electronic grounds. This happened despite the fact that the average trend line for chemical and physical properties v Z going down B-Al-Sc-Y... Ac is more regular.
In main group IV, notice how C and SI are positioned in the middle of the cell, unlike their neighbours to either side. The group thus bifurcates after Si into a Ti branch and a Ge branch. This is quite reasonable since there is not much difference in the average trendlines going down either option. In any case, C-Si came to be moved over Ge again on electronic grounds.
He survived the electronic revolution, staying over Ne.
| Year: 1921 | PT id = 1237, Type = formulation |
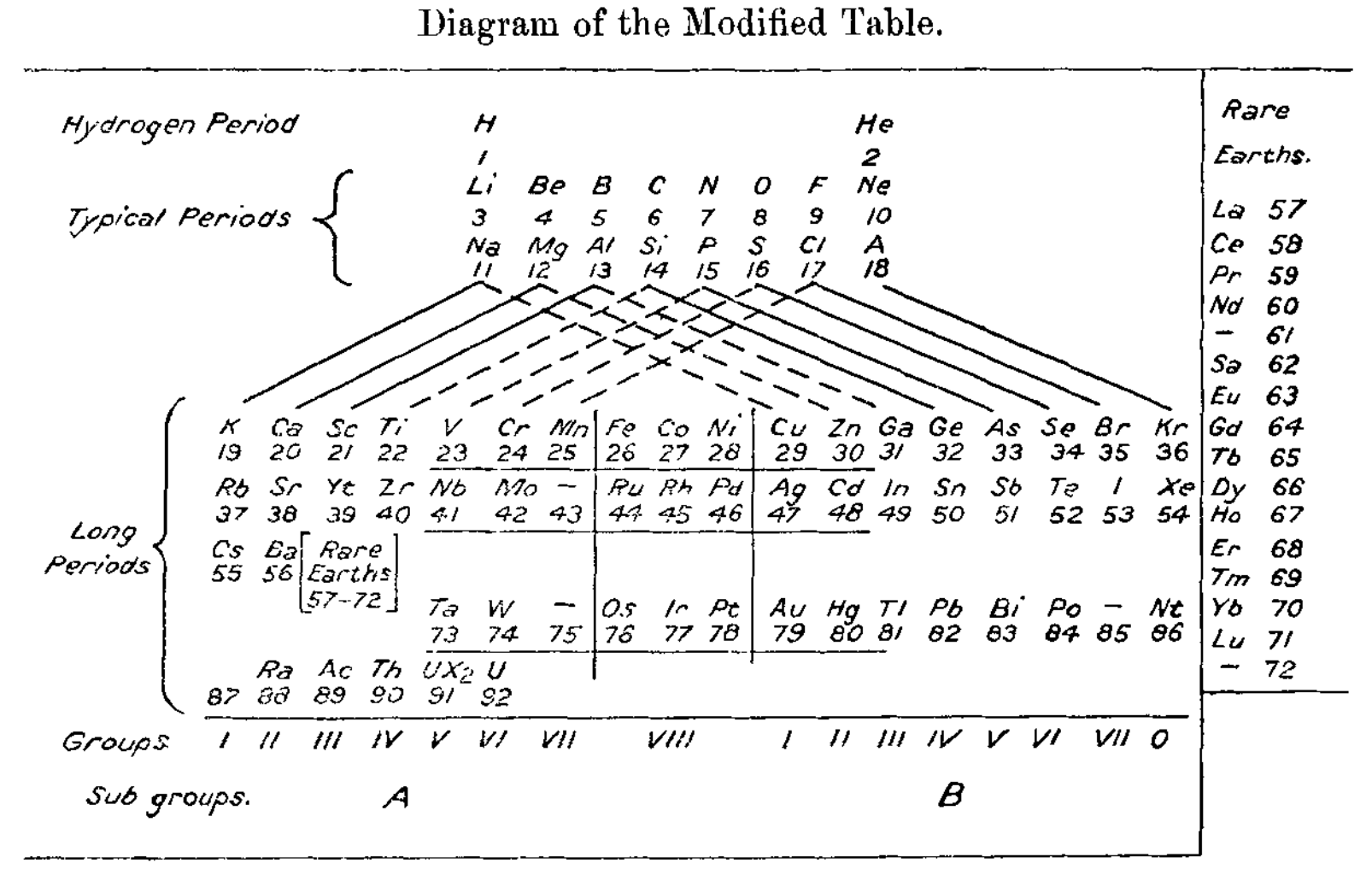
Margary's Modified Table
Ivan D. Margary B.A. (1921) XXXVI. A modification more in accord with atomic structure, The London, Edinburgh, and Dublin Philosophical Magazine and Journal of Science, 42:248, 287-288.
An old school table showing group 3 as B-Al-Sc-Yt-Rare earths.
Thanks to René for the tip!
 |
 |
 |
| What is the Periodic Table Showing? | Periodicity |
© Mark R. Leach Ph.D. 1999 –
Queries, Suggestions, Bugs, Errors, Typos...
If you have any:
Queries
Comments
Suggestions
Suggestions for links
Bug, typo or grammatical error reports about this page,please contact Mark R. Leach, the author, using mark@meta-synthesis.com
This free, open access web book is an ongoing project and your input is appreciated.